The Q-Box CO650 Plant CO2 Analysis Package has been designed for measurements of photosynthesis, respiration and transpiration in leaves when placed in a flow-through leaf chamber. You can use Plant CO2 Analysis Package in both the lab or field (with optional battery pack). The Q-Box CO650 software automatically checks the reference levels of CO2 and water vapour and provides on-the-go calculations of photosynthesis and transpiration rates.
- Modular Gas Exchange Measuring System
- Flow-through leaf chamber
- Components interchangeable with other Q-Box Packages
- Automated calculations of carbon and water exchange rates
- Housed in rugged box for easy transport and set up
- Use in a lab or Field (with optional battery pack)
- Photosynthetic CO2 Exchange Rate measurements
- Transpiration Rate measurements
- Water Use Efficiency studies
- Dark Respiration Rate measurements
- Leaf, Root and Whole Plant Studies (with optional chambers)
- Soil Respiration or animal respiration with additional components
- Q-A101 laboratory stand (free standing and/or integrated into Q-Box)
- A113 LED Light Source Calibrated in Software
- Q-G267 Flow Monitor, Gas Pump x 2 (1L/min no load)
- G112 Flow Through Leaf Chamber
- G122 Large Gas Bags (2)
- Q-S151 CO2 Analyzer (0-2000ppm) (Includes CO2 and H2O scrubbers)
- Q-S161 RH/temperature sensor
- S173 Leaf Thermister
- A383 DCU Solenoid for switching between reference and sample gas
- S181 Absolute Pressure Sensor
- C610 Three integrated LabQuest Mini data interfaces (9 Analog Channels)
- C901 Logger Pro Software
- C404 Customized Experimental Setup Files for collection, display and analysis of data
- Q-Box Accessory Kit (tubing, filters, connectors)
- Rugged Water-proof case housing the sensors and analyzers
- Manual
- individual power supplies for stand alone use of the sensors and analyzers
- Li-ion 4 Battery pack (optional)
- Iseas MS et al (2025) Influence of the microtopography of patagonian peatbogs on the fluxes of greenhouse gasses and dissolved carbon in porewater. Ecohydrology & Hydrobiology 25, 155-165, https://doi.org/10.1016/j.ecohyd.2024.01.013
- Rudresh HS et al (2024) Comparative analysis of desiccation tolerance in Oesporangium elegans and Mickelopteris cordata. Vegetos 37: 1244-1256 https://doi.org/10.1007/s42535-023-00664-8
- Petrova S, Petkova M. (2023) Plant Traits of Tilia tomentosa Moench, Fraxinus excelsior L, and Pinus nigra J.F. Arnold as a proxy of urbaniazation. Forests 14, 800 https://doi.org/10.3390/f14040800
- Oconor EF et al (2023) Growth and photosynthetic development of Swietenia mahagoni in svbstrates inoculated with arbuscular mycorrhizal fungi. Bosque 44, https://doi.org/10.4067/S0717-92002023000200329
- Marriboina S et al. (2020) Systematic hormone-metabolite network provides insights of high salinity tolerance in Pongamia pinnata (L.) pierre doi: https://doi.org/10.1101/2020.04.28.066050
- Yathisha NS et al. (2020) Vegetative desiccation tolerance in Eragrostiella brachyphylla: biochemical and physiological responses. Heliyon 6, Cell Press Online https://doi.org/10.1016/j.heliyon.2020.e04948
- Sengupta D et al. (2019) Photosynthetic performance and sugar variations during key reproductive stages of soybean under potassium iodide-simulated terminal drought. Photosynthetica 57: 458-469
- Jacotot, A et al. (2018) Effects of elevated atmospheric CO2 and increased tidal flooding on leaf gas-exchange parameters of two common mangrove species: Avicennia marina and Rhizophora stylosa. Photosynth Res 138, 249–260. https://doi.org/10.1007/s11120-018-0570-4
- Van de Poel B et al (2016) Transcriptome Profiling of the Green Alga Spirogyra pratensis (Charophyta) Suggests an Ancestral Role for Ethylene in Cell Wall Metabolism, Photosynthesis, and Abiotic Stress Responses. Plant Physiology 172 533-546
- Tian L et al. (2015) Research on the Effect of Electrical Signals on Growth of Sansevieria under Light-Emitting Diode (LED) Lighting Environment. PLoS ONE 10(6):1-18 DOI:10.1371/journal.pone.0131838
- Eisingera W. et al. (2012) Microtubules Are Essential for Guard-Cell Function in Vicia and Arabidopsis. Molecular Plant 5: 601-610

An LED Light Source (A113) supplies photosynthetically active radiation to the leaf with minimum heat load. The LED light source can deliver approximately 2000 µmol photons/m2/s at maximum output and it is supplied calibrated (in the software). As a result light intensity can be accurately set without the use of a light meter. For calculations of leaf conductance, leaf temperature measurements are required and these are obtained by the S173 Leaf Thermistor that is fitted in the bottom portion of the flow-through leaf chamber (G112). Analog signals from all of the sensors are converted to digital signals via three integrated LabQuest mini interfaces (9 channels). Data is displayed, recorded and manipulated on a PC or Macintosh computer using Logger Pro software. Each package comes with customized experimental files for data collection, automated calculations and any further analysis.
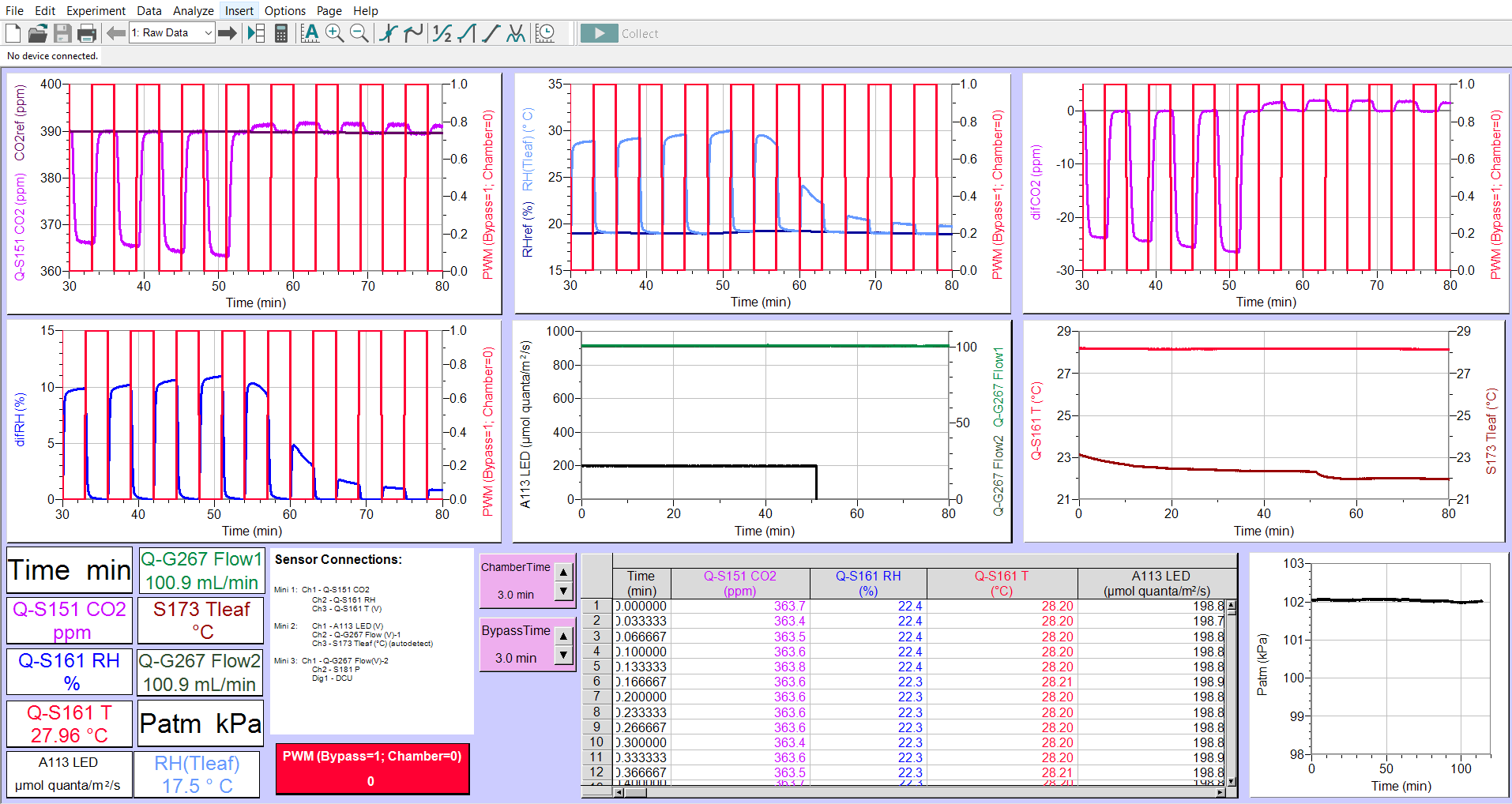
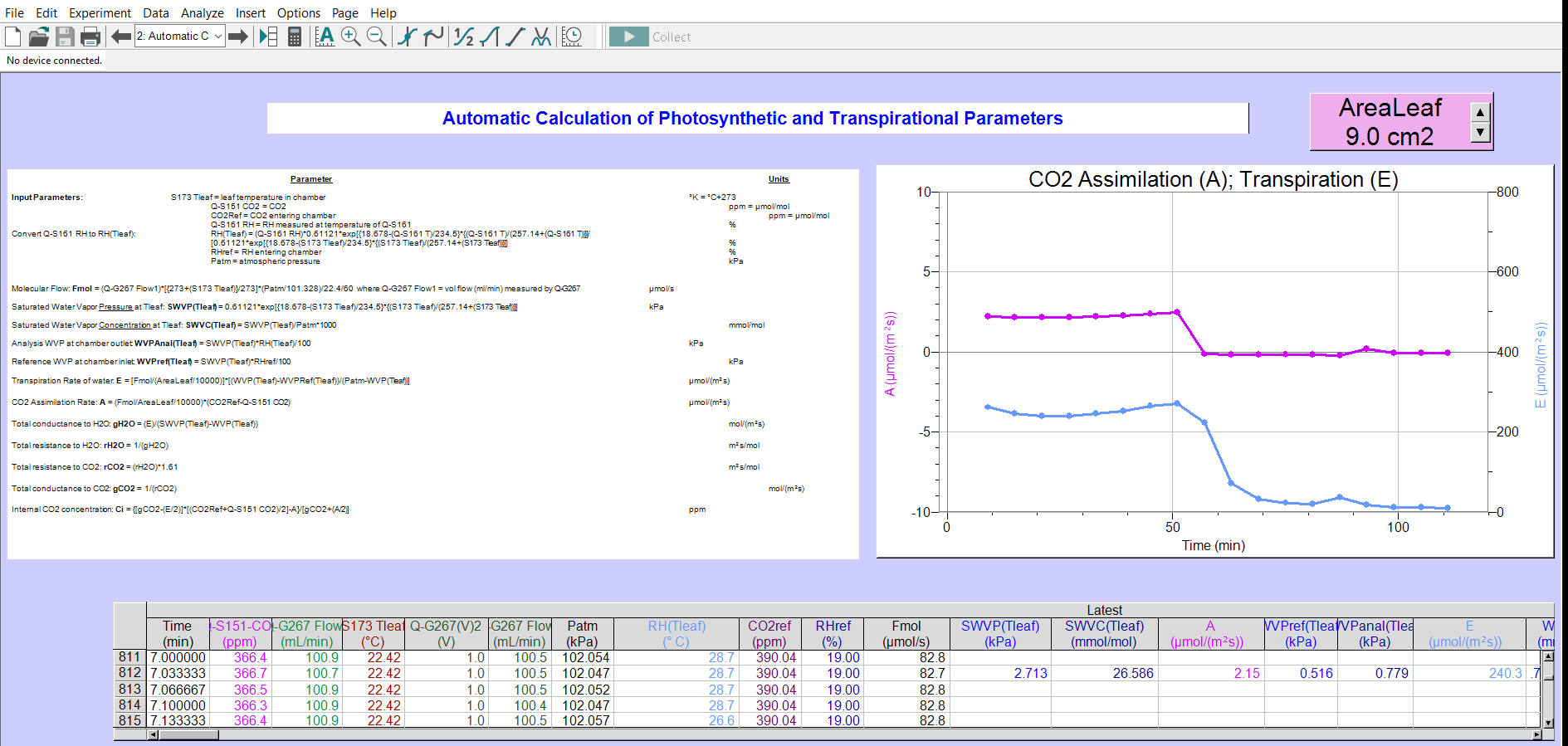
Operation of the Q-Box CO650 Plant CO2 Analysis Package employs the use of one infrared CO2 gas analyzer (Q-S151). The concentration of CO2 gas entering a leaf chamber and exiting it, is measured at different times with the same analyzer. User specified control of the DCU solenoid valve assembly switches between reference and sample measurements. When the leaf is exposed to light the difference between influx and efflux CO2 levels (differential CO2), plus measurement of the gas flow through the chamber, allow automatic calculation of photosynthetic CO2 fixation rate. Measurements of CO2 exchange in the dark provide data of dark respiration.
The Q-Box CO650 includes a Humidity/Temperature sensor (Q-S161) which measures relative humidity of the air before and after it has passed through the leaf chamber. At the same time, temperature at the RH sensor is also measured. The RH differential between influx and efflux gas, the temperature, the flow rate through the leaf chamber and leaf temperature are used in automatic calculation of leaf transpiration rates.
Q-Box CO650 Software
Testimonial
“We first started using the Qubit photosynthesis package shortly after I began teaching Plant Physiology in 1998. Initially, we purchased one package, and it worked so well that we soon added three more. Since that time, my class has used the system for two labs each semester including one that deals with photosynthesis and one that deals with respiration. During this time, we only had to upgrade the original sensors once…so their durability and long-lasting performance is remarkable. So I was able to get nearly 13 years of use from the initial packages that I purchased….even with hundreds of students using them! Their system is very student friendly, and performs very well in a classroom situation. Students are excited to see real-time measurements, and the software allows them to calculate rates very easily. Hence, the quality of their product is truly exceptional.``
Dr. Janice M. Coons, Professor of Botany, Biological Sciences Department, Eastern Illinois University, Charleston, IL